What is OHDSI?
OHDSI (pronounced "Odyssey") is a large, open-science initiative dedicated to bring out the full value of observational health data through standardized data, standardized analytics, and building a global community.
The OHDSI community brings together a diverse, international network of experts and users across disciplines such as clinical medicine, epidemiology, biostatistics, computer science, and life sciences. OHDSI consists of researchers, healthcare providers, patients, regulators, and industry partners – all working together to generate reliable, reproducible, scalable real-world evidence.
At its core, OHDSI aims to maximize the potential of real-world data by addressing key challenges in health research, including limited data interoperability and reproducibility. To overcome these barriers, OHDSI develops and adopts the Observational Medical Outcomes Partnership Common Data Model (OMOP CDM) to harmonize the structure and content of observational health data. This facilitates a federated data network, enabling large, multi-database studies while preserving patient privacy and respecting local data governance standards.
Key Focus Areas
- Data Standardization - Promoting consistent data structure and analytics through the OMOP CDM and open-source tools.
- Safety Surveillance - Detecting and evaluating potential adverse effects of medical products using large-scale, real-world data.
- Comparative Effectiveness - Comparing treatment options to understand what works best in real-world settings.
- Personalized Risk Prediction - Using patient-level data to estimate individual health risks and support decision-making.
- Data Characterization - Assessing data quality and understanding data sources to ensure reliable research.
- Quality Improvement - Supporting health systems with evidence-based tools to measure and improve care.
How the OHDSI Approach Works
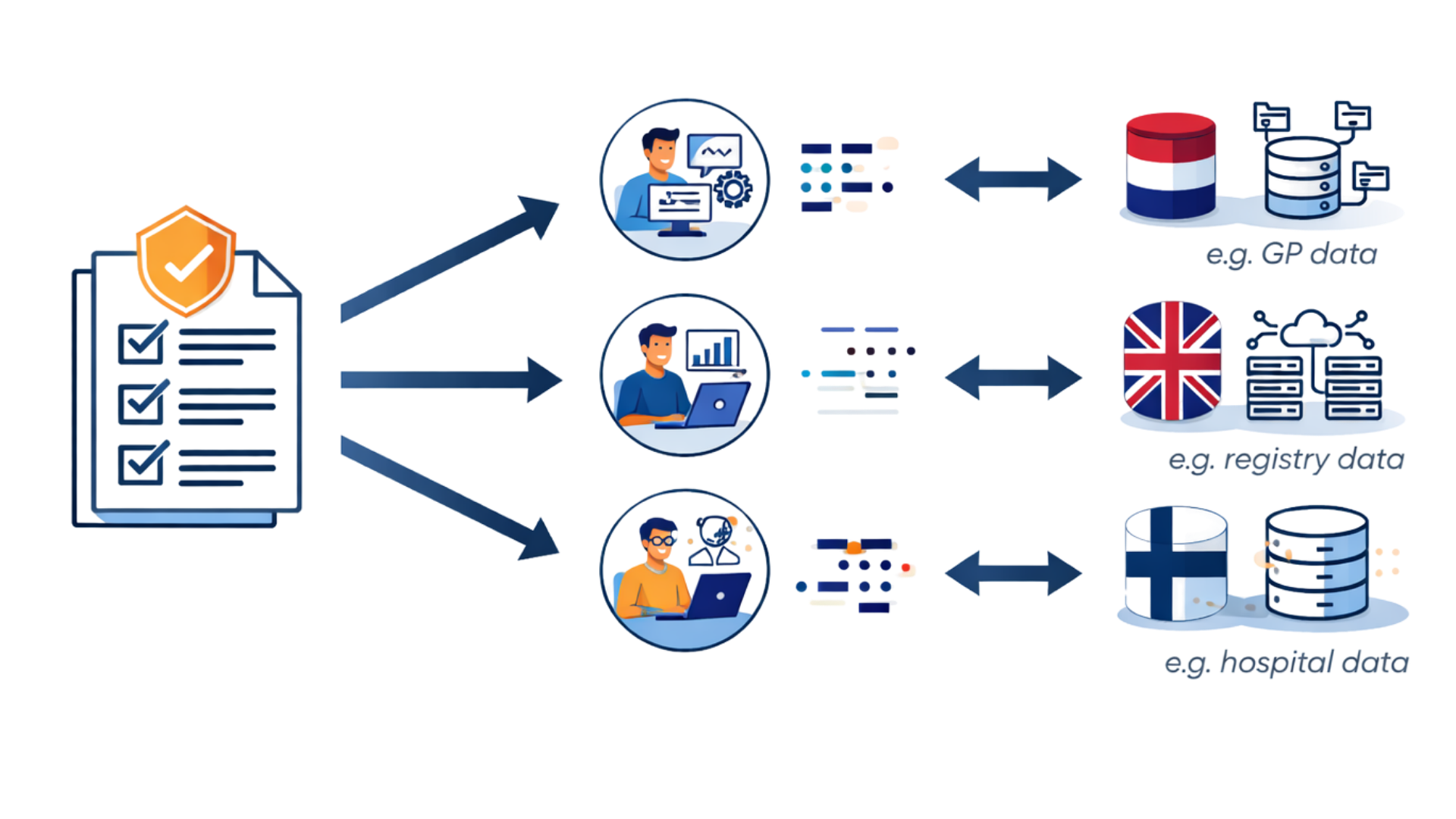
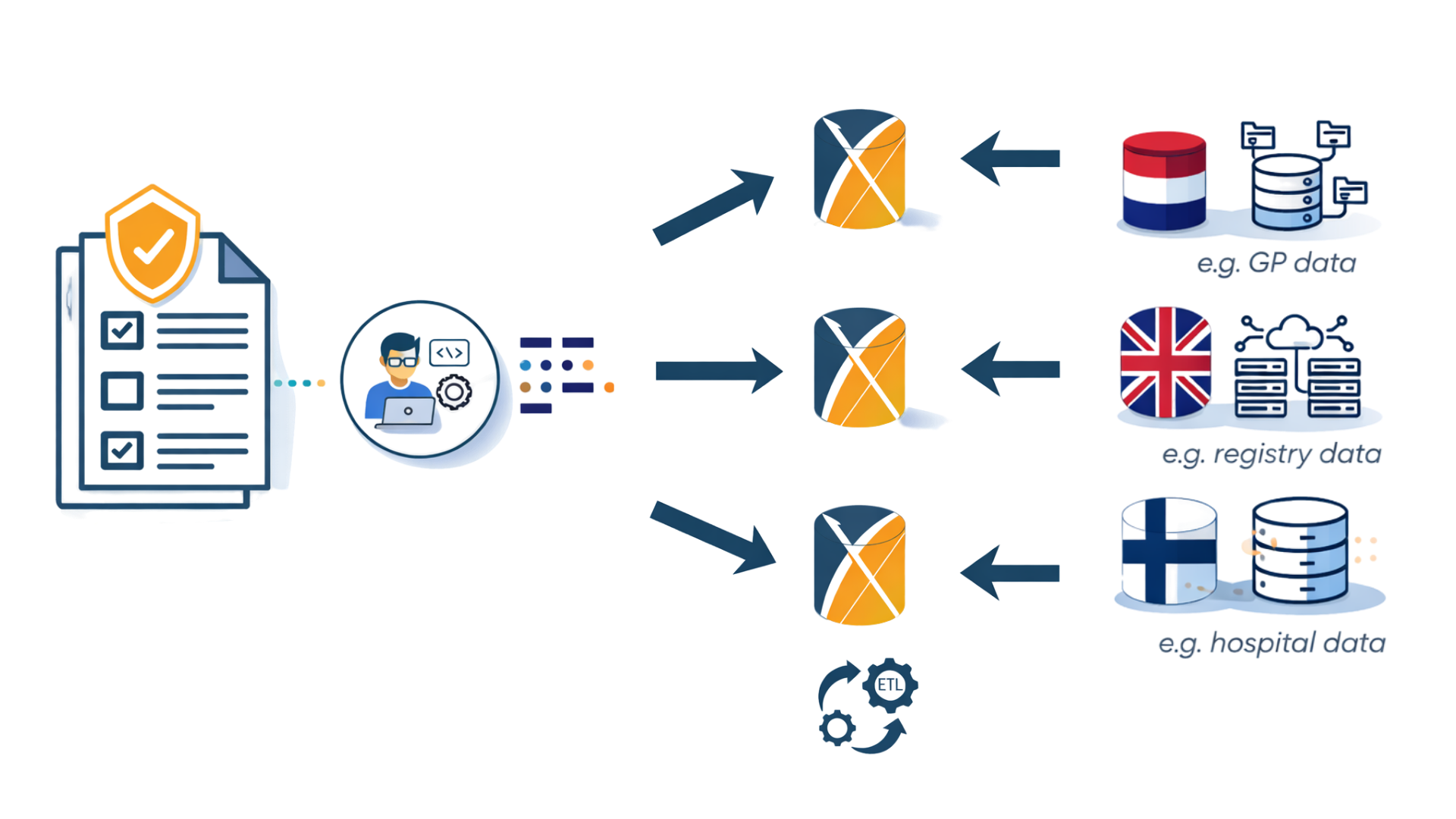
Why the OHDSI Approach Works
OHDSI enables federated analyses across observational health databases worldwide. Studies can be executed locally and results combined, without sharing patient-level data—supporting robust evidence across countries, populations, and care settings.
Standardized data and reusable analytical pipelines reduce the time and effort needed to answer new clinical and regulatory questions—enabling reliable evidence to be generated quickly when it matters most.
Open-source tools built around a common data model allow studies to be replicated and run across multiple databases and diseases—improving transparency, consistency, and long-term scientific value.
The Key Building Blocks of OHDSI
Standardized Data
The OMOP CDM harmonizes diverse sources such as electronic health records, claims data, and registries into a single, consistent structure.
Standardized vocabularies and OMOP tables enable both semantic and syntactic interoperability, making heterogeneous real-world data analyzable with the same phenotypes and analytical tools.
Standardized Analytics
OHDSI provides a comprehensive suite of open-source tools that support the full observational research lifecycle — from ETL and data quality assessment to cohort definition, clinical characterization, patient-level prediction, and population-level effect estimation.
ATLAS offers a user-friendly interface for common analyses on OMOP CDM data without programming skills. For advanced and customizable workflows, HADES provides a collection of R packages designed for reproducible and scalable analytics on OMOP CDM databases.
All OHDSI tools are freely available and continuously developed by the community.
A Global Community
OHDSI is powered by an active international community of researchers, clinicians, and data partners. Collaboration across institutions and countries strengthens study quality, accelerates learning, and avoids working in isolation.
Join a workgroup, attend our annual symposium, or connect with the community in your country by joining your national node.
